SIH COVID Update
Last Update - 5/12/2023
Masking Requirements in SIH Patient Care Facilities
Per CDC guidelines, community transmission levels of COVID-19 determine whether masks are required in healthcare facilities. When transmission levels are “low to substantial” in the county the facility is located, masking is not required. When the facility is in a county with “high” transmission levels, masks remain a requirement. For the latest community transmission levels, by county, click here.
SIH represents four hospitals and many clinical sites across our seven-county primary service area. We will monitor transmission levels for each of our seven counties and post those metrics here on Fridays.
Counties reporting “high” transmission (Masks required in all patient care areas):
Counties reporting “low to substantial” transmission (No mask required for most patients and visitors *):
- Franklin, Jackson, Johnson, Perry, Randolph, Saline, Union, Williamson
* Patients and visitors will still be required to wear a mask if:
- COVID positive or suspected to have COVID or any other respiratory virus.
- Have had close contact with someone positive for COVID within the last 10 days.
SIH COVID-19 Results by Calendar Day with Daily % Positive and Rolling Average
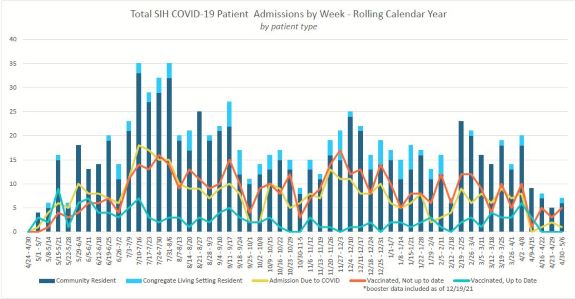
Total SIH COVID-19 Patient Admissions by Week - Rolling Calendar View By Patient Type
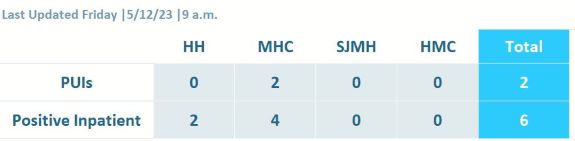
SIH COVID-19 Current Inpatient Data
Patient Under Investigation (PUI) - An inpatient who is awaiting test results.
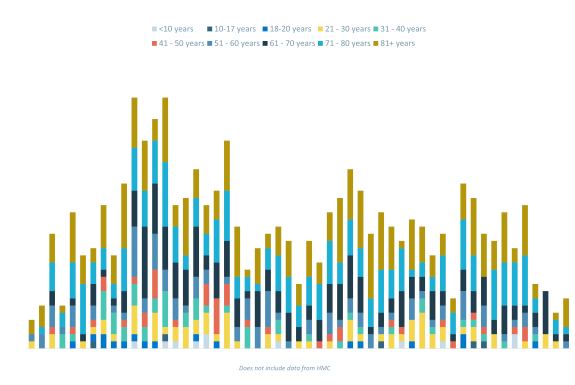
SIH COVID-19 Admission By Age
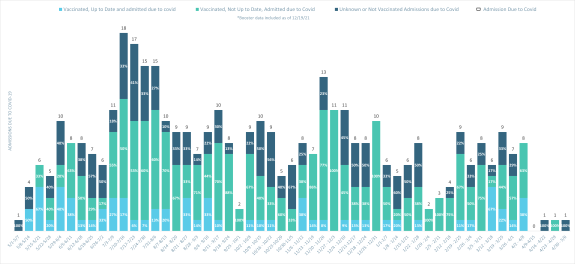
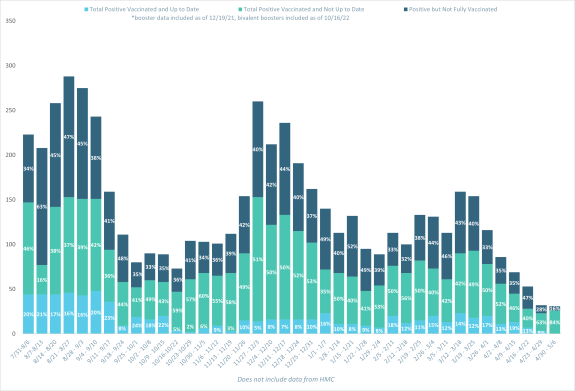
Inpatient Admissions due to COVID-19 with Vaccination Status by Week
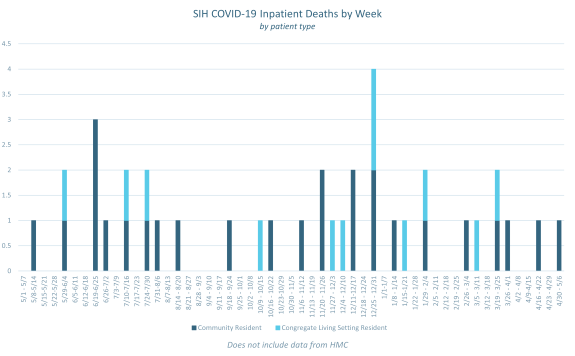
Inpatient Deaths due to COVID-19 with Vaccination Status by Week
SIH Total Number of Fully Vaccinated Breakthrough COVID-19 Cases
This chart includes testing data from SIH COVID-19 testing locations and hospitals.
COVID-19 Risk Metrics and Vaccination in Southern Illinois
A county enters a warning level when it experiences an increase in two or more of the COVID-19 risk indicators from the state’s COVID-19 Resurgence Mitigation plan. A map and information of each county’s status can be found on the IDPH website at https://www.dph.illinois.gov/countymetrics
State & National COVID-19 Data
COVID-19 Reminders
Vaccine Fact Sheets
- Johnson & Johnson Fact Sheet for Recipients and Caregivers
- Moderna Fact Sheet for Recipients and Caregivers
- Pfizer Fact Sheet for Recipients and Caregivers
Travel Guidelines
For information from the CDC on travel recommendations for both fully-vaccinated and unvaccinated individuals, click here. Fully vaccinated travelers are less likely to get and spread COVID-19.
To learn when it is advisable to avoid or delay travel, click here. Upon returning to work from travel, please continue to follow all entry screening processes and comply with the universal masking policy in all facilities.
Donning & Doffing
- Video: Donning and Doffing for COVID 19_CAPR
- Video: Donning and Doffing PPE for COVID-19 with procedure mask
- Video: How to Properly Don & Doff an Ear-Loop Facemask
HIPAA
Every patient has a right to privacy. Inappropriate access, use or disclosure of patient information is a minimum of a Level 3 Improvement Counseling under our Confidentiality of Sensitive Information Policy.
Community Information COVID-19 Quick Reference
SIH Community COVID-19 Hotline
Call 844.988.7800
8am - 4:30pm, 7 Days a Week
COVID-19 Vaccine Information & Scheduling
Call 866.744.2468
8am - 4:30pm, 7 Days a Week
How to get tested
Covid-19 testing will be done at SIH Walk-In Clinics. Online Scheduling will no longer be needed.
- Testing will be offered as part of an office visit if a provider determines it is necessary.
- For travel or other testing needs, you may wish to visit a local pharmacy.